Let's Talk About Electrolytes
The minerals running the show inside your body
Happy April!
Spring may have sprung, but you can already feel summer elbowing its way in. The mornings are warmer, the days are longer, and nature is basically begging you to come outside and sweat. Speaking of which, that is exactly where we are headed in today’s newsletter.
On a personal note: the end of the spring semester is finally in sight, and I am so close to the finish line I can practically taste it. Between rotations, coursework, side gigs, and conference planning, I have been running on caffeine, conviction, and, doing my best to stay hydrated. In my Micronutrient course, we just began discussing electrolytes, and the timing felt too perfect not to share. So here we are.
Today we are breaking down electrolytes from the ground up: what they actually are, why your body needs them, who needs to pay extra attention, and how much is actually enough. No fluff, just real science made digestible (see what I did there?).
So, What Even is an Electrolyte?
Let's start at square one. The word "electrolyte" sounds intimidating, but the concept is beautifully simple. When certain minerals dissolve in a liquid like your blood or the fluid inside your cells, they split into tiny charged particles called ions. Some carry a positive charge, some carry a negative charge. That charge is the key.
Think of it like this: your body runs on tiny electrical signals that tell your muscles to contract, your heart to beat, and your brain to fire. Electrolytes are the conductors of that electricity. Without them, nothing moves.
The main minerals are sodium, potassium, magnesium, calcium, chloride, phosphate, and bicarbonate. You have heard of most of them, as they show up in your food, your blood work, and yes, on the label of that little packet of Liquid IV or LMNT you may or may not have tried.
Electrolytes are not a wellness trend: they are foundational to life itself, as they are essential for maintaining electrical neutrality in cells and generating the action potentials that power your nerves and muscles.
The Main Players
Sodium: regulates fluid outside cells, drives thirst and supports nerve signals.
Potassium: regulates fluid inside cells, heart rhythm, and muscle contractions.
Magnesium: supports 300+ enzyme reactions, energy production and sleep.
Calcium: supports bone health, muscle contractions, and nerve transmissions.
Chloride: balances fluid, aids digestion (as part of stomach acid).
Bicarbonate: keeps blood pH in the sweet spot (around 7.4)
Sodium and potassium work as a famous duo. Because sodium dominates outside your cells, while potassium handles business inside, together they operate something called the sodium-potassium pump, a tiny molecular machine that maintains the voltage across your cell membranes. That voltage is what makes nerve signaling and muscle contraction physically possible. Every time you take a step on a treadmill, that pump is firing millions of times a second.
How You Lose Them
Your body loses electrolytes in a few key ways: through urine (constantly, in small amounts), through sweat (especially during exercise or in heat), and through vomiting or diarrhea when you are sick. Your kidneys are the main regulators. They filter excess electrolytes out through urine and hold onto them when your levels dip low.
Sweat is mostly water, but it also carries sodium and chloride, which is why dried sweat sometimes leaves a white residue on your skin or workout gear. If you are a salty sweater, you already know. Potassium, magnesium, and calcium are lost in smaller amounts through sweat, but over long, hard efforts they add up.
Not Everyone’s Needs are the Same
For most healthy adults eating a reasonably balanced diet, electrolyte needs are met through food. But certain groups genuinely require more intentional replenishment.
Athletes and active people: Anyone doing vigorous exercise for more than an hour, especially in heat, is losing meaningful amounts of sodium and other electrolytes through sweat. Endurance athletes can lose up to 2 to 3 quarts of fluid per hour during intense activity. That is not just water walking out the door.
People working outdoors: Construction workers, farmers, landscapers, and anyone spending long hours in the sun face the same risk as athletes when it comes to sweat losses. The difference is they might not be thinking about it.
Kids: Children have a higher body surface area relative to their size, so they lose fluids and electrolytes faster than adults do. When kids are sick with vomiting or diarrhea, dehydration can escalate quickly.
Older adults: The sense of thirst becomes less reliable with age, and kidney function changes, meaning older adults are more vulnerable to imbalances without realizing it.
People on certain medications: Diuretics, the medications that increase urination, can significantly deplete sodium, potassium, and magnesium. If you or someone you care for is on a diuretic, electrolyte monitoring is often part of standard clinical care.
How Much Do You Actually Need?
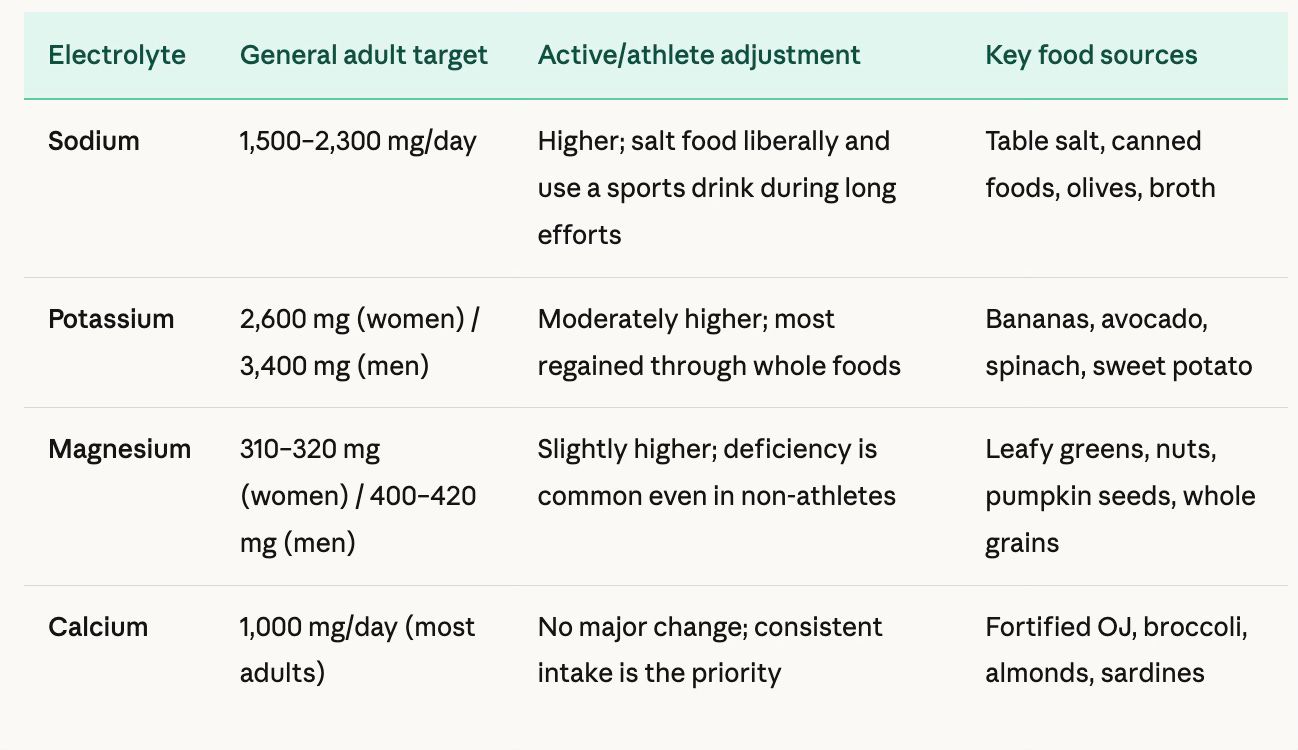
Here is a breakdown of general daily recommendations based on current Dietary Reference Intakes (DRIs) and clinical guidance. Keep in mind these are population level guidelines. Individual needs vary based on body size, sweat rate, health conditions, and activity level.
For athletes specifically, sports science research suggests that electrolyte drinks become genuinely useful when exercise exceeds about 60 minutes, particularly in warm or humid conditions. A good benchmark is to aim for a beverage with at least 150 to 180 mg of sodium per 8 oz serving. The Johns Hopkins dietetics team puts it plainly: in sports hydration, sodium is the star, not potassium. Coconut water, while trendy and tasty, is high in potassium but relatively low in sodium, making it a less complete choice for post-sweat replenishment on its own.
What Imbalance Looks Like?
Electrolyte imbalances are more common than most people realize, and they can range from mildly annoying to genuinely serious. Muscle cramps, fatigue, brain fog, headaches, and dizziness are early warning signs that something may be off. In severe cases, imbalances can cause heart arrhythmias, seizures, or in the case of extreme low sodium, confusion and coma.
Low potassium (hypokalemia) is one of the most commonly seen imbalances in clinical settings, often showing up as muscle weakness and irregular heartbeats. Low magnesium is particularly sneaky, and perhaps the most common of the electrolyte deficiencies. It can cause twitching, anxiety, poor sleep, and even contribute to low calcium because magnesium plays a role in how the body regulates parathyroid hormone.
The good news is for most healthy people eating a varied diet and staying reasonably hydrated, your body is remarkably good at keeping electrolytes in balance on its own. The kidneys are the unsung heroes, constantly filtering, retaining, and excreting based on what you need.
The Takeaway
For the general population, you probably do not need to be counting milligrams of sodium or taking powdered electrolyte supplements daily. Eat a variety of whole foods, drink water throughout the day, and listen to your body.
Where it gets more important is during extended exercise (especially in heat), illness involving vomiting or diarrhea, or if you are in a high-risk group like older adults or people on certain medications. In those situations, reaching for an electrolyte drink, oral rehydration solution, or even a salty snack with water is a smart move.
And as summer creeps in, because it absolutely is, those sweat heavy outdoor runs and backyard workouts are going to ask more of your body’s electrolyte reserves. Consider this your advance notice to plan accordingly.
Until next time: drink water, lightly salt your food, and go outside while the weather is nice.
Ever Well,
Megan Hysaw